28 Day
The Sleep Study with INDI Rest
| STUDY TITLE | The Sleep Study with INDI Rest |
|---|---|
| Submitted under umbrella | |
| Date submitted |
Aug 11, 2025 |
| End date |
There is no pre-specified end date and sub-studies remain open long-term. |
| Language |
English |
| Efforia AI IRB approval recommendation | |
| Recruitment page preview | |
| Author edit | |
| Informed consent |
|
| Study author |
Chris Read, Matthew Amsden |
| Principal investigator |
Matthew Amsden |
| Sub-Investigator for Adverse Events |
Dr. Viral Patel |
| Description |
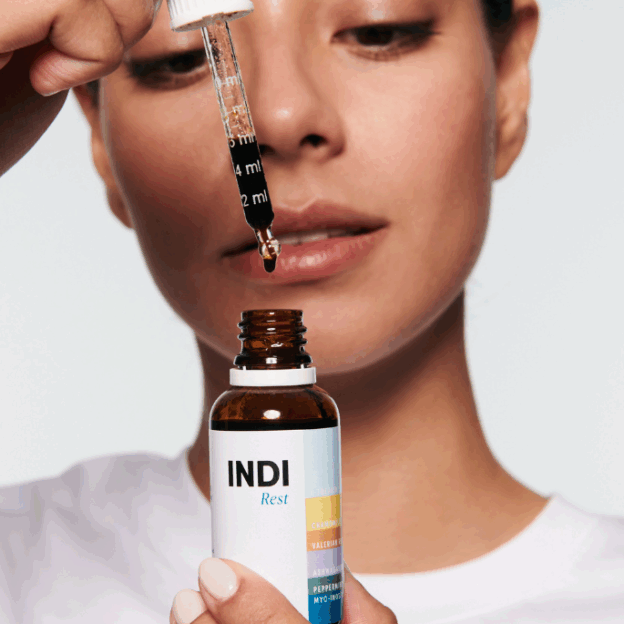
Can plants really help you sleep? With 90% of adults reporting sleep issues at some point, it's impact affects peoples health and productivity worldwide. INDI Rest harnesses the most effective all-natural ingredients to optimise your sleep. |
| Participant engagement length |
28 Days |
| Sponsor |
This study is made possible by your payment to cover all supplies and expenses required to participate. |
| Cost to participant |
$15 |
| Included products & services |
INDI – Rest Trial: $20
|
| Outcome measures |
PROMIS Item Bank v.1.0 – Sleep-Related Impairment Survey PROMIS Sleep Disturbance Scale |
| Methodology |
Single Arm longitudinal where participants act as their own control |
| Basic or advanced dissemination plan |
Basic |
| Deviation from recruitment approach |
No |
| Deviation from statistical approach |
No |
| Will study include “more about you questions” |
No |
| Clinicaltrials.Gov |
Yes |
| Committment to list findings on clinicaltrials.Gov |
Yes |
This document is prepared with the assistance of AI, but is reviewed by a human.
Rational & Study Design
The Intervention
Included Products & Services
Product Name: INDI – Rest Trial
Quantity included: 0
Price: $20.00
Product Description: Natural sleep remedy The most potent combination of natural ingredients available to ease anxiety, dispel racing thoughts and induce deep and restful sleep: 89% of trial participants saw an improvement in sleep in just one week, and not a single person felt groggy the day after use.
Product Image:
Ingredients:
Product Safety:
Study Design & Methodology
Inappropriate Participants & Inclusion/Exclusion
Under the Efforia minimal risk protocol, within which this study is reviewed for safety and human subjects ethics, all individuals over the age of consent are eligible, however, it is the responsibility of participants to determine if their unique circumstances make participation inappropriate.
Here is a table identifying the kinds of people who should avoid being part of the INDI Rest trial:
| Category of Individual | Contraindications | Reason |
|---|---|---|
| Individuals who drive or operate heavy machinery | INDI Rest may cause drowsiness | The intervention's effects could impair alertness and reaction times, increasing the risk of accidents. |
| Individuals on other medications | Potential for interaction with natural ingredients | Although rare, there could be interactions between INDI Rest and other medications that participants are taking. |
| Individuals with certain medical conditions | Consult with a doctor if concerned | Some medical conditions could be exacerbated by the ingredients or effects of INDI Rest. |
| Pregnant or breastfeeding individuals | Lack of robust studies in these populations | The effects on pregnancy and breastfeeding are unknown, and caution is advised. |
| Individuals with a history of severe allergies | Potential for allergic reactions to ingredients | Natural ingredients could trigger allergic reactions in sensitive individuals. |
| Individuals with sleep disorders | Need for professional diagnosis and treatment | Sleep disorders may require more comprehensive interventions than INDI Rest can provide. |
Study Design & Experience
Statistical Analysis Plan
Limitations & Justification
The single-arm observational design of this study, conducted under the Minimal Risk Citizen Science Umbrella Protocol, inherently presents several limitations, primarily the potential for bias due to the absence of a control group. Without a comparator, it becomes challenging to definitively attribute changes in sleep quality directly to the intervention, INDI Rest. However, to address this limitation, we have implemented an expectations questionnaire to quantify the potential bias introduced by participants' expectations. By understanding the baseline expectations, we can better interpret the results and adjust our findings accordingly, providing a more nuanced understanding of INDI Rest's impact on sleep quality.
Additionally, the study's sample size and statistical methodology, including potential stratification by participant demographics, have been thoughtfully crafted to further mitigate bias. By examining various demographic factors, we aim to ensure that the findings are not unduly influenced by specific participant characteristics. Although the absence of a randomized control group remains a limitation, our design leverages the participants as their own controls, utilizing standardised measures such as the Pittsburgh Sleep Quality Index (PSQI) to track improvements over the study period. This strategy, while not eliminating bias, helps in yielding preliminary insights into the efficacy of INDI Rest, thereby serving as a valuable signal detection study.
Furthermore, the overarching goal of Efforia and the Minimal Risk Umbrella Protocol to democratize clinical research underpins our study design. By focusing on research questions and researchers that might otherwise be overlooked, this study not only seeks to validate the claims surrounding INDI Rest but also aims to empower participants with personalized insights into their sleep quality. Should the signal be positive, we are committed to pursuing more robust study designs to replicate and validate these initial findings in a controlled setting. This iterative approach ensures that any potential benefits identified are thoroughly examined, reinforcing the integrity and applicability of the research outcomes. For more detailed information on the Minimal Risk Umbrella Protocol, interested individuals are encouraged to consult the relevant documentation.
Human Subjects Ethics
Suitability Under Minimal Risk Umbrella Protocol
The protocol titled "The Sleep Study with INDI Rest" appears to be suitable for the minimal risk umbrella protocol based on the criteria provided. This study likely focuses on examining the effects of INDI Rest, which is presumably a non-regulated dietary supplement or a consumer health device aimed at enhancing sleep quality. The study aligns with the umbrella protocol's focus on interventions related to general health, productivity, and wellness within normal ranges. Assuming INDI Rest is classified as a "Generally Recognized as Safe" (GRAS) intervention and does not require a prescription or physician diagnosis, it fits well within the framework provided. Additionally, if the study is structured as a longitudinal study over several days and involves minimal bodily fluid specimen collection, it adheres to the guidelines for minimal risk studies.
However, for the protocol to be entirely suitable, it must ensure that it does not involve vulnerable populations, require any kind of medical device or regulated pharmaceutical intervention, or include more than two blood draws throughout the study duration. Furthermore, it should avoid any form of blinding, deception, or comparative study design. Provided these conditions are met, and the study adheres to examining legal, accessible interventions in its jurisdiction, "The Sleep Study with INDI Rest" would be a suitable candidate under the minimal risk umbrella protocol. It must also ensure that all participants are of legal consenting age and that the study does not include any biased or unnecessary elements outside the scope of improving health and wellness.
Suitability for Pay to Participate Model
In considering the appropriateness of allowing participants to pay for the INDI Rest Trial in "The Sleep Study with INDI Rest," it's essential to evaluate the product's availability and market acceptance. INDI Rest, which utilizes all-natural ingredients to optimize sleep, is legally available on the open market in the countries where the study will be conducted. People experiencing sleep issues, a common problem affecting 90% of adults at some point, are generally willing to pay for solutions that promise improved sleep quality and well-being, making this product relevant and appealing to the target demographic. The pricing of $15 for the study is relatively modest and competitive, as similar products in the health and wellness market often come at a similar or higher cost. This pricing ensures that typical consumers can access the trial without experiencing serious financial hardship, supporting equitable participation.
Furthermore, the value proposition of the INDI Rest Trial extends beyond just the convenience of purchasing a sleep aid. The study offers participants the opportunity to contribute to scientific knowledge about natural sleep optimization, providing an additional layer of value through data-driven insights into their sleep patterns and overall health benefits. The voluntary nature of participation, combined with comprehensive informed consent and transparency about the product and study outcomes, helps mitigate any potential for undue influence or buyer's remorse. By empowering participants with evidence-based health decisions, the study aligns with ethical principles of respect for persons, beneficence, and justice, ensuring a responsible and beneficial research experience.
Human Subjects Protection Questionnaire
Beneficence
Is there a description, unambiguous research question, and purpose? Yes, the study aims to investigate the effectiveness of a specific product or service, detailing its purpose and expected outcomes for participants. This provides clarity and direction for the research.
Is the study built on what is known already? Yes, the study leverages existing knowledge and previous research to inform its methodology and objectives, ensuring it contributes constructively to the field.
Will the study provide meaningful answers to the research question? Yes, the study is designed to yield results that are both generalizable and relevant to individual participants, thereby offering insights that can enhance understanding and application of the product or service.
Will the study provide valid answers to the research question? Yes, the study employs a rigorous methodology to ensure that the findings are credible and can be confidently used to draw conclusions about the research question.
Non-maleficence
Are participants recruited with justifiable inclusion and exclusion criteria? Yes, any individual who has already purchased the product is eligible to participate, and they are informed of the minimal risks and special considerations through the consent process.
Does the research team have the experience, skills, facilities, and time to complete the study? Yes, the decentralized nature of the study requires minimal patient interaction, and Efforia has established extensive infrastructure. The team, led by PI Matthew Amsden, is well-equipped to manage the study, with Dr. Viral Patel available to address any adverse events.
Is there a fair balance of benefits and harms (risks) for all with an interest in the study? Yes, the study's design under the minimal risk protocol offers value to participants at no additional cost, creating a balance between benefits and potential risks.
Will participants receive appropriate care both during and after the study? Yes, participants are informed to seek their own medical care for any adverse events. The study offers more oversight than typical market use, particularly in monitoring for adverse events.
Is personal data handled appropriately (confidentiality)? Yes, personal data is managed in accordance with the guidelines set out in the Minimal Risk Umbrella protocol.
Autonomy
Have participants been offered a fair choice through the information they are given (presented in plain English) and consent process? Yes, the informed consent document provides detailed information about the study, enabling participants to make informed decisions about their involvement.
Has the research incorporated patient and participant views? Yes, the study is participant-driven, allowing for feedback and input from participants throughout the study, aligning with Efforia's citizen science approach.
Justice
Are there fair payments for participation and financial recompense in case of harm? Yes, while participants pay to participate, they receive a valuable product and tracking of health outcomes. In case of harm, participants are responsible for their medical expenses but can report incidents to Efforia.
Do participants have access to an independent complaints procedure (or advocate)? Yes, participants can contact Efforia's support for concerns, and independent ethics review board contacts are included in the consent.
Will the project be registered and results reported in the public domain? Yes, Efforia ensures transparency, providing immediate personal results to participants and contributing generalizable results to the public domain.



