
30 Day
30 Day Insulin Balance Metabolic Health Optimization Protocol
| STUDY TITLE | 30 Day Insulin Balance Metabolic Health Optimization Protocol |
|---|---|
| Submitted under umbrella | |
| Date submitted |
Oct 18, 2025 |
| End date |
There is no pre-specified end date and sub-studies remain open long-term. |
| Language |
English |
| Efforia AI IRB approval recommendation | |
| Recruitment page preview | |
| Author edit | |
| Informed consent |
|
| Study author |
Elwin Robinson |
| Principal investigator |
Matthew Amsden |
| Sub-Investigator for Adverse Events |
Dr. Viral Patel |
| Description |
Explore how a dietary supplement can help support healthy insulin levels, and it's impact on weight, fatigue, and sugar cravings. The Insulin Balance Formula and Hormetics Weight Optimization System is designed for people who are generally healthy who want to support and maintain healthy insulin levels to reach general health goals. Join the study and complete periodic measures to see how the product helps you reach your health goals. |
| Participant engagement length |
30 Days |
| Sponsor |
This study is made possible by your payment to cover all supplies and expenses required to participate. |
| Cost to participant |
$49 |
| Included products & services |
Insulin Balance Formula – Hormetics Weight Optimization System – 1 bottle: $49
|
| Outcome measures |
PROMIS Sleep Disturbance Scale Body Composition Measurement Form After Meal Hunger & Fatigue Perception Carbohydrate Craving Questionairre |
| Methodology |
Single Arm longitudinal where participants act as their own control |
| Basic or advanced dissemination plan |
Basic |
| Deviation from recruitment approach |
No |
| Deviation from statistical approach |
No |
| Will study include “more about you questions” |
No |
| Clinicaltrials.Gov |
Yes |
| Committment to list findings on clinicaltrials.Gov |
Yes |
This document is prepared with the assistance of AI, but is reviewed by a human.
Rational & Study Design
Why I created this study:
With insulin at the heart of metabolic health, the dance between diet, weight, and energy has captivated both scientific and popular discourse. While there are many tools for individuals with diabetes and significant obesity issues, there's less research on tools to help individuals who otherwise have insulin levels in a healthy range, but still have carb cravings, after meal fatigue and mood swings.
My Objective for You:
Our quest is to empower you with the knowledge of how a dietary supplement might support optimal insulin levels within a healthy range - and how this might impact weight, fatigue, and cravings. By the end of this study, you'll have a personalized understanding on how the supplement worked for you. This isn't just about data—it's about giving you the tools to optimize your health and make informed decisions about your lifestyle choices. Please note, this study is not designed to diagnose or treat any kind of insulin disorder.
Aims & Objectives:
We aim to evaluate the effects of the Insulin Balance Formula on insulin levels, weight, fatigue, and sugar cravings in healthy adults. Participants will gain personalized insights into the product's effectiveness, enabling them to make informed decisions about their health and wellness journey.
Significance & Impact:
This study offers an opportunity to scrutinize the intricate relationship between insulin regulation and overall metabolic health. While we anticipate positive outcomes in weight management and reduced cravings, limitations include individual variability and adherence to the protocol. Nonetheless, the potential to influence personal health strategies and contribute to broader nutritional science makes this study a groundbreaking endeavor.
The Intervention
Included Products & Services
Product Name: Insulin Balance Formula – Hormetics Weight Optimization System – 1 bottle
Quantity included: 30
Price: $49.00
Product Description: The Insulin Balance Formula is for the 'High Insulin Type' who is struggling with weight loss due to insulin resistance, where cells become less responsive to insulin, leading to elevated blood sugar and fat storage. A cornerstone of the Hormetics Weight Optimization System, this product is doctor-formulated by Dr. Miriam Mikicki, MD and Elwin Robinson.
Product Image:
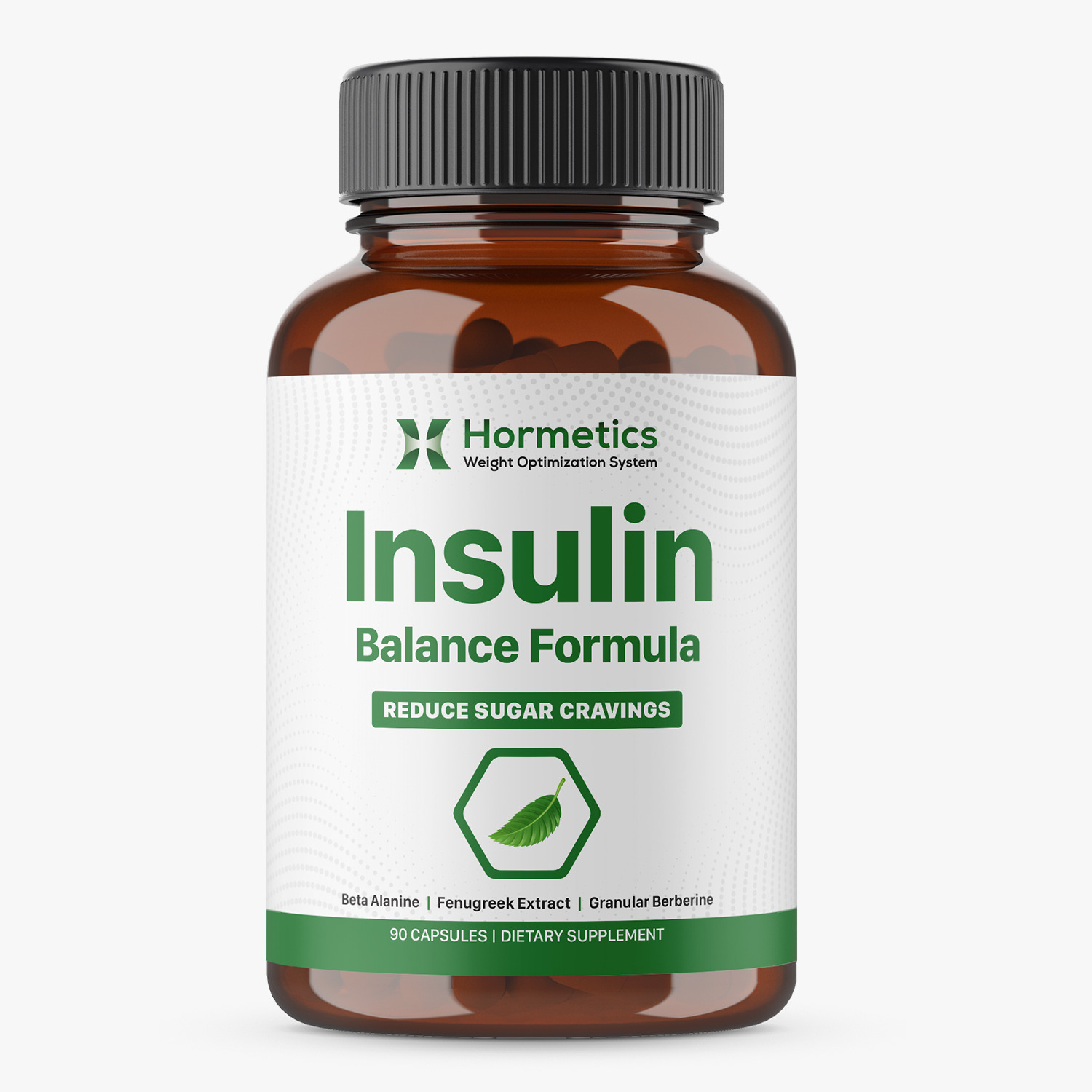
Ingredients:
Who This Is For
The Insulin Balance Formula is crafted for individuals struggling with weight loss due to insulin resistance, where cells become less responsive to insulin, leading to elevated blood sugar and fat storage [2]. This doctor-formulated solution is perfect for:
Adults with stubborn weight gain, particularly around the midsection [3].
Those with metabolic syndrome or prediabetes seeking to enhance insulin sensitivity [4].
Individuals desiring a natural, doctor-formulated approach to hormonal balance and weight management [5].
If diets have failed you or you feel stuck in a cycle of weight gain, this doctor-formulated formula offers a targeted path to reclaim your health.
Benefits of Insulin Balance Formula
Hormetic’s Insulin Balance Formula delivers a range of benefits to support your weight loss and overall wellness:
Enhances Insulin Sensitivity: Improves your body’s response to insulin, reducing blood sugar spikes and fat storage [6].
Promotes Sustainable Weight Loss: Supports fat burning by correcting hormonal imbalances with a doctor-formulated approach [7].
Reduces Cravings: Stabilizes blood sugar to curb sugar cravings and overeating [8].
Supports Metabolic Health: Promotes healthy cholesterol, blood pressure, and energy levels [9].
Optimizes Hormonal Balance: Aligns with the Hormetics Weight Optimization System for comprehensive wellness [10].
Each ingredient is selected for synergy, delivering powerful results backed by science.
Our Ingredients: Science Meets Tradition
The Insulin Balance Formula, doctor-formulated by Dr. Miriam Mikicki, MD, and Elwin Robinson, combines five potent ingredients, each chosen for its proven ability to combat insulin resistance and support weight loss. Sourced from premium suppliers, these ingredients blend ancient wisdom with modern science:
Berberine 500mg
What It Is: Berberine is a bioactive compound extracted from plants like Berberis vulgaris (barberry) [11].
Source: Sustainably harvested from high-quality botanical sources, our Berberine ensures potency in this doctor-formulated formula [12].
History of Use: Used for over 2,000 years in Chinese and Ayurvedic medicine, Berberine is celebrated for its metabolic benefits [13].
Benefits: A 2012 study found Berberine reduced BMI from 31.5 ± 3.6 to 27.4 ± 2.4 kg/m² and leptin levels from 8.01 to 5.12 μg/L in metabolic syndrome patients [14]. This doctor-formulated ingredient activates AMPK, promoting weight loss and insulin sensitivity [15].
Picture of berberine berries
Beta Alanine 500mg
What It Is: Beta Alanine is a non-essential amino acid known for buffering acid in muscles and enhancing metabolic function [16].
Source: Synthesized from natural precursors, our pharmaceutical-grade Beta Alanine ensures purity in this doctor-formulated formula [17].
History of Use: Traditionally used in sports nutrition, Beta Alanine has emerged as a key player in metabolic health [18].
Benefits: A 2021 study found Beta Alanine enhances insulin sensitivity and reduces oxidative stress in metabolic syndrome patients [19]. This ingredient supports energy metabolism, helping you stay active and burn fat [20].
Zinc Citrate 15mg
What It Is: Zinc Citrate is a highly absorbable form of zinc, an essential mineral for hormonal and metabolic health [21].
Source: Sourced from natural mineral deposits, our chelated Zinc Citrate optimizes absorption in this doctor-formulated blend [22].
History of Use: Zinc has been used for centuries in traditional medicine to support immunity and hormonal balance [23].
Benefits: A 2010 study showed zinc improves insulin sensitivity and reduces fasting glucose in insulin-resistant individuals [24]. This ingredient supports healthy lipid profiles and reduces inflammation, aiding weight loss [25].
Folate (L-5-MTHF) 200mcg
What It Is: L-5-Methyltetrahydrofolate (L-5-MTHF), the bioactive form of folate, is a B-vitamin crucial for DNA synthesis and metabolic health [26].
Source: Derived from natural plant-based precursors, our folate is highly bioavailable, ensuring maximum efficacy in this doctor-formulated blend [27].
History of Use: Folate has been used for decades to support cellular health, with recent research highlighting its role in metabolic disorders [28].
Benefits: A 2021 study showed folate improves insulin sensitivity and reduces homocysteine levels in insulin-resistant patients [29]. This ingredient supports blood sugar regulation and reduces inflammation, aiding weight management [30].
Fenugreek Extract 500mg
What It Is: Fenugreek Extract, derived from Trigonella foenum-graecum seeds, is rich in bioactive compounds like saponins [31].
Source: Organically grown and standardized, our Fenugreek ensures consistent potency in this doctor-formulated blend [32].
History of Use: Fenugreek has been used for centuries in Middle Eastern and Indian traditions to support metabolism [33].
Benefits: A 2015 study showed Fenugreek reduces fasting glucose, insulin, and body weight in prediabetic patients [34]. This ingredient stabilizes blood sugar and reduces cravings with its high fiber content [35].
[3] https://pmc.ncbi.nlm.nih.gov/articles/PMC4591578/,
[4] https://pubmed.ncbi.nlm.nih.gov/34371867/,
[5] https://pubmed.ncbi.nlm.nih.gov/34333586/
[6] https://pubmed.ncbi.nlm.nih.gov/22474499/,
[7] https://pmc.ncbi.nlm.nih.gov/articles/PMC4591578/,
[8] https://pubmed.ncbi.nlm.nih.gov/34333586/,
[9] https://pubmed.ncbi.nlm.nih.gov/20045801/,
[10] https://pubmed.ncbi.nlm.nih.gov/34371867/
[11] https://pubmed.ncbi.nlm.nih.gov/22474499/,
[12] https://pubmed.ncbi.nlm.nih.gov/23118793/,
[13] https://pubmed.ncbi.nlm.nih.gov/23118793/,
[14] https://pubmed.ncbi.nlm.nih.gov/22474499/,
[15] https://pubmed.ncbi.nlm.nih.gov/22851982/
[16] https://pubmed.ncbi.nlm.nih.gov/34333586/,
[17] https://pubmed.ncbi.nlm.nih.gov/21847611/,
[18] https://pubmed.ncbi.nlm.nih.gov/21847611/,
[19] https://pubmed.ncbi.nlm.nih.gov/34333586/,
[20] https://pubmed.ncbi.nlm.nih.gov/21847611/
[21] https://pubmed.ncbi.nlm.nih.gov/20045801/,
[22] https://pubmed.ncbi.nlm.nih.gov/17292847/,
[23] https://pubmed.ncbi.nlm.nih.gov/17292847/,
[24] https://pubmed.ncbi.nlm.nih.gov/20045801/,
[25] https://pubmed.ncbi.nlm.nih.gov/19107981/
[26] https://pubmed.ncbi.nlm.nih.gov/34371867/,
[27] https://pubmed.ncbi.nlm.nih.gov/17209196/,
[28] https://pubmed.ncbi.nlm.nih.gov/28724658/,
[29] https://pubmed.ncbi.nlm.nih.gov/34371867/,
[30] https://pubmed.ncbi.nlm.nih.gov/28724658/
[31] https://pmc.ncbi.nlm.nih.gov/articles/PMC4591578/,
[32] https://pubmed.ncbi.nlm.nih.gov/21947880/,
[33] https://pubmed.ncbi.nlm.nih.gov/21947880/,
[34] https://pmc.ncbi.nlm.nih.gov/articles/PMC4591578/,
[35] https://pubmed.ncbi.nlm.nih.gov/16379570/
Product Safety:
Who Shouldn’t Take This Formula
While the Insulin Balance Formula is safe for most adults, certain individuals should avoid it or consult a healthcare provider before using this product:
Pregnant or Nursing Women: The safety of Berberine and Fenugreek during pregnancy or breastfeeding is not established [36].
Individuals on Certain Medications: Berberine may interact with medications like metformin or statins [37].
Those with Low Blood Sugar: This formula may lower blood sugar, so caution is advised for hypoglycemic individuals [38]. Taking the capsules with a meal may help.
People with Allergies: Those allergic to fenugreek or related plants (e.g., peanuts) should avoid this product [39].
Children: This doctor-formulated formula is not intended for individuals under 18 [40].
Consult your healthcare provider before starting this supplement, especially if you have health conditions or take medications.
Study Design & Methodology
Inappropriate Participants & Inclusion/Exclusion
Under the Efforia minimal risk protocol, within which this study is reviewed for safety and human subjects ethics, all individuals over the age of consent are eligible, however, it is the responsibility of participants to determine if their unique circumstances make participation inappropriate.
Here is a table identifying the kinds of people who should avoid being part of this trial based on potential risks and the intervention:
| Category of Individual | Contraindications | Reason |
|---|---|---|
| Individuals with Known Allergies | Possible allergic reactions to ingredients | Risk of allergic reactions and skin/hair reactions based on known side effects. |
| Pregnant or Nursing Individuals | Lack of safety data | Limited research on supplement's effect on pregnancy or lactation. |
| Individuals with Insulin Disorders | Supplement not intended to treat insulin disorders | Could lead to improper management of insulin levels. |
| Individuals with GI Disorders | Gastrointestinal symptoms | Known risk of exacerbating gastrointestinal symptoms. |
| Individuals with Mental Health Conditions | Mood changes and mental health discomfort | Potential exacerbation of mood changes and mental health discomfort. |
| Individuals with Sleep Disorders | Sleep disturbances | Risk of worsening sleep patterns due to known sleep disturbances. |
| Individuals on Medication | Potential interactions | Risk of interaction with existing medications, potentially altering effects. |
| Athletes under Specific Regulations | Legality for use | Possible conflicts with regulations of sports organizations. |
| Individuals Sensitive to Fatigue | Known fatigue side effect | Risk of increased fatigue impacting daily activities. |
These categories are based on potential risks and existing health conditions that may be exacerbated by participating in the study. Participants should consider these factors and consult with their healthcare providers.
Study Design & Experience
Daily Activity: Take 3 capsules of the Insulin Balance Supplement daily, ideally 1 capsule before each meal or all at once for convenience. Begin with 1 capsule to assess tolerance and increase to the recommended dose. Take on an empty stomach or with a meal if needed.
Assesments and frequency:- Body Composition Measurement: Complete the assessment as required.
- PROMIS Sleep Disturbance Scale: Complete the assessment as required.
- Carbohydrate Craving Questionnaire: Complete the assessment as required.
- After Meal Hunger & Fatigue Perception: Complete the assessment as required.
The "Insulin Balance Metabolic Health and Weight Optimization Protocol" is an observational study designed to investigate the effects of a dietary supplement on insulin levels and its subsequent impact on weight management, fatigue, and cravings for sugar. In this study, participants engage as their own control in a single-arm method, a scientific approach known as the "Signal Phase" at Efforia. This trial aims to detect initial positive outcomes that suggest the supplement's effectiveness in promoting metabolic health. Through this phase, not only will potential positive results be identified, but it will also gather critical safety data that is currently limited, thereby paving the way for more comprehensive studies in the future.
Participants will partake in a 30-day structured protocol, during which they will incorporate the Insulin Balance Formula into their daily routines alongside a balanced diet. The study is crucial as it addresses the challenges faced by many in maintaining stable insulin and blood sugar levels, which are essential for metabolic health. By closely monitoring their response to the supplement, participants will provide valuable insights into how this nutritional strategy may enhance energy levels, improve mood consistency, and support healthy weight maintenance. Their role is pivotal in assessing the formula's impact under real-world conditions, generating meaningful data that can inform future interventions.
Throughout the study, participants are encouraged to engage actively and provide honest feedback, ensuring the collection of accurate and personalized results. The study emphasizes the importance of privacy and data confidentiality, assuring participants that their information will only be used within the research context. Participants are also advised against introducing new supplements or health interventions during the trial to avoid confounding results. Should any health concerns arise, they are instructed to seek medical attention immediately. The insights gained from this study could significantly influence the understanding of nutritional support in managing blood sugar levels, potentially leading to more robust research designs if positive signals are detected. For a comprehensive overview, reviewers are encouraged to consult the "Minimal Risk Umbrella protocol" for detailed information.
Expected sample size:Thank you for considering our submission for IRB review. We want to emphasize that our statistical plan is consistent with the Minimal Risk Umbrella protocol under which this study is categorized. The Minimal Risk Umbrella protocol provides a comprehensive framework that ensures the safety and ethical integrity of studies that pose minimal risk to participants, such as ours.
For reviewers who wish to assess our statistical plan in detail, we recommend reviewing it within the context of the Minimal Risk Umbrella protocol. This umbrella protocol outlines the statistical methodologies, risk assessments, and data management strategies that are applicable to all studies under its purview, including the Insulin Balance Metabolic Health and Weight Optimization Protocol. By aligning with this umbrella, we ensure our study adheres to established guidelines and maintains methodological consistency.
Should you have any specific questions regarding how our study's statistical plan aligns with the umbrella protocol, we are more than willing to provide additional clarifications. We appreciate your attention to our submission and look forward to your feedback.
Statistical Analysis Plan
All similar studies at 30, 60 and 90 will be analyzed together.
Limitations & Justification
This single-arm observational trial aims to explore the effects of the Insulin Balance Formula on insulin levels, weight, fatigue, and sugar cravings in healthy adults. However, the study design inherently lacks a control group, which can introduce bias and limit the generalizability of the findings. Recognizing this limitation, we have implemented an expectations questionnaire to quantify and understand participant biases. This tool allows us to assess the influence of participant expectations on the outcomes, thereby providing a clearer interpretation of how the Insulin Balance Formula might genuinely impact metabolic health. By addressing the bias through structured assessment, we aim to enhance the reliability of our findings, despite the inherent limitations of a single-arm study.
To mitigate further limitations, the study's sample size and statistical methodologies are carefully crafted, including potential stratification by participant demographics. This stratification allows for a nuanced analysis of how different demographic factors might influence the outcomes, thereby providing a more comprehensive understanding of the supplement's effects. Additionally, the study is designed as a signal detection trial, meaning that its primary goal is to identify potential signals of efficacy that warrant further investigation. Should a positive signal be detected, subsequent studies with more robust designs, possibly including randomized controlled trials, can be initiated to replicate and validate the initial findings. This iterative approach ensures that any potential benefits are thoroughly tested in a more controlled and rigorous setting, thus addressing the initial study's limitations over time.
A central tenet of the Minimal Risk Citizen Science Umbrella Protocol, under which this study is conducted, is to democratize clinical research. This means supporting research questions and researchers that might otherwise be overlooked. By focusing on healthy adults who experience carb cravings, after-meal fatigue, and mood swings—yet maintain insulin levels within a healthy range—this study aims to fill a gap in existing research. The study empowers participants with personalized insights and contributes to broader nutritional science, emphasizing practical health optimization strategies over disease treatment. For more information on how the Minimal Risk Umbrella Protocol facilitates these research endeavors, we encourage individuals to refer to the detailed guidelines provided by Efforia.
Human Subjects Ethics
Suitability Under Minimal Risk Umbrella Protocol
At first glance, the title “Insulin Balance Metabolic Health and Weight Optimization Protocol” might appear to suggest a medical intervention or treatment for insulin-related disorders. However, this study and product clearly fall within the parameters of Efforia’s Minimal Risk Umbrella Protocol based on the following considerations:
-
Purpose and Population:
This study is not designed to diagnose, treat, or prevent any insulin disorder, nor is it intended for individuals with abnormal glucose or insulin function. Rather, it is intended for generally healthy adults with normal insulin levels who wish to optimize their metabolic function, energy, and body composition—similar to how healthy consumers use over-the-counter continuous glucose monitors to track and understand their responses to meals and exercise. -
Regulatory Classification:
All ingredients in the Insulin Balance Formula are classified as dietary supplement ingredients that are generally recognized as safe (GRAS) under U.S., EU, and U.K. regulations. None require a prescription, and none are restricted or regulated as drugs. The supplement therefore qualifies as a low-risk, legally marketed, non-prescription product appropriate for consumer use. -
Risk Profile:
The study procedures involve daily supplement use and simple self-reported measures, all of which are non-invasive and minimal in risk. There are no blood draws, device implants, or pharmacological interventions.
Accordingly, while the name “Insulin Balance” may imply a medical context, the actual design, intent, and regulatory classification of the product and study place it squarely within the scope of Efforia’s Minimal Risk Umbrella Protocol.
Suitability for Pay to Participate Model
In evaluating the Insulin Balance Metabolic Health and Weight Optimization Protocol, it is appropriate to allow participants to pay for the products and services included in the study. The Insulin Balance Formula and Hormetics Weight Optimization System are products that are generally available legally on the open market in the United States. Consumers who are interested in supporting healthy insulin levels and managing their weight are typically willing to invest in dietary supplements and health optimization systems, often paying similar or even higher prices for comparable health products. The price of $49 for the one month supply represents a competitive offering, providing participants with a clear and tangible product that aligns with their health goals.
Moreover, the study offers additional value beyond just the consumption of the products. Participants receive the benefit of engaging with a structured protocol that includes periodic measures to track and evaluate their progress, which offers a personalized experience and insight into how the products affect their health goals. The typical target market for these products—individuals who are health-conscious and focused on maintaining or improving their health—can generally afford such investments without experiencing serious financial hardship. Furthermore, the study is designed for individuals who are generally healthy, reducing the risk of severe and acute participant vulnerabilities that might lead to significant buyer's remorse. Therefore, the pay-to-participate model in this study is ethically justified, supporting both participant autonomy and the integrity of the research.
Human Subjects Protection Questionnaire
Beneficence
Is there a description, unambiguous research question, and purpose? Yes, the study includes a clear research question and purpose, aiming to evaluate the effects of a specific product on health outcomes. This clarity ensures that the study is focused and directed towards obtaining meaningful insights.
Is the study built on what is known already? Yes, the study builds upon existing knowledge by leveraging prior research and data on the product's effects. This foundation allows the study to explore new aspects and deepen understanding within the context of previous findings.
Will the study provide meaningful answers to the research question? Yes, the study is designed to provide meaningful answers not only by generating generalizable results but also by offering insights pertinent to individual participants' experiences with the product. This dual approach enhances the study's relevance and impact.
Will the study provide valid answers to the research question? Yes, the study utilizes a robust methodology to ensure that the answers obtained are valid. This includes appropriate data collection techniques and analysis methods that are aligned with the research question.
Non-maleficence
Are participants recruited with justifiable inclusion and exclusion criteria? Yes, the inclusion criteria are clear, allowing any individual who has already purchased the product to participate. Efforia emphasizes participant responsibility to self-assess their suitability for the minimal risk protocol through detailed informed consent.
Does the research team have the experience, skills, facilities, and time to complete the study? Yes, the research team is well-equipped with the necessary experience and infrastructure to conduct the study. Efforia's platform supports decentralized trials, and the Principal Investigator, Matthew Amsden, ensures expert oversight. Dr. Viral Patel is available to handle any adverse events, reinforcing the study's safety.
Is there a fair balance of benefits and harms (risks) for all with an interest in the study? Yes, the study's minimal risk protocol offers added value to participants who have already purchased the product, without additional cost. This approach ensures a fair balance between potential benefits and risks.
Will participants receive appropriate care both during and after the study? Yes, participants are advised to seek their own medical care in the event of an adverse reaction, as outlined in the informed consent. The study, being of minimal risk, provides participants with more care and monitoring than they would typically receive outside of the research setting.
Is personal data handled appropriately (confidentiality)? Yes, personal data is handled in accordance with the standards set by the Minimal Risk Umbrella protocol, ensuring confidentiality and privacy.
Autonomy
Have participants been offered a fair choice through the information they are given (presented in plain English) and consent process? Yes, the informed consent document is written in plain English and offers comprehensive information, allowing participants to make an informed decision about their involvement in the study.
Has the research incorporated patient and participant views? Yes, the study is participant-driven, reflecting Efforia's commitment to citizen science. Participants are encouraged to provide feedback and input, ensuring their views are incorporated throughout the study.
Justice
Are there fair payments for participation and financial recompense in case of harm? Yes, while participants pay to participate, they receive a valuable product and health outcome tracking, providing intrinsic value. Participants are informed that medical care is their financial responsibility, and they are instructed to report adverse events to Efforia.
Do participants have access to an independent complaints procedure (or advocate)? Yes, participants can contact Efforia's support for any complaints, with the option to reach out to an independent ethics review board as detailed in the informed consent.
Will the project be registered and results reported in the public domain? Yes, Efforia ensures transparency by making personal results immediately available to participants and aims to publish generalizable results to build credibility.



